Indira Gandhi National Open University (IGNOU) 2005 B.Sc Chemistry Organic Reaction Mechanism - - Question Paper
CHE-6
BACHELOR OF SCIENCE (B.Sc,) Term-End Examination December, 2005
CHEMISTRY CHE-6 : ORGANIC REACTION MECHANISM
Time : 2 hours Maximum Marks : 50
Note : Attempt any four questions. At! quest tons carry equal marks.
1. (a) Arrange the following halides in the increasing order
of their reactivity towards SN2 reaction : 1~
C6H5CH2Br; CH2 = CH CH2Br; CHgBr
(b) What is isotopic labelling ? How is it used to probe
the mechanism of a reaction ? Explain with example. 3
(c) Why is 2-chloroethyi ethyl sulphide hydrolysed in acetone 10,000 times faster than the corresponding ether ? 3
(d) (i) Nitrobenzene upon nitration with fuming HNOg/H2SO mixture forms m-dinitrobenzen as the major product. Explain with mechanism. 3
(ii) Why are alkenes more reactive than alkynes
towards electrophilic addition reactions ? 2
2. (a) What are stereospecific and stereoselective reactions ?
Explain with one example in each case. aL
2
(b) Write a detailed note on any two of the following
rearrangements : 8
(i) Pinacol-pinacolone rearrangement
(ii) Benzil-benzilic acid rearrangement
(iii) Hofmann rearrangement
3. (a) What is the product obtained when neopentyl bromide
is heated with dilute ethanol ? Write all the steps involved. qJ.
(b) Give structures and names of the two isomeric products obtained when CgH5CH2CHClC6H reacts with alcoholic KOH. Give mechanism of the reaction. 3
(c) Give mechanism for the reaction of propene with N-bromosuccinimide. 3
(d) What are singlet and triplet carbenes ? Draw their structures. 3
(i) Fropanone from diethyl malonate
(ii) Acetyi acetone (pentane-2,4-dione) from ethyl acetoacetate
(iii) p-bromoaniline from aniline
(b) Write short notes on any three of the following : 2x3
(i) Fluorescence
(it) Photosensitiseition
(iii) Radical anion
(iv) Intersystem crossing
(c) Arrange following radicals in the increasing order of their stability ; jL
2
.ch3, ,ch(Gh3)2, .C(CH3)3, .ch2ch3
5. (a) Complete the following reactions :
6
|
0) (CH3)3CCHC1 A B |
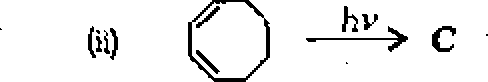 |
+ CH2(COOC2Hs)2
CsT
CgHsQNa D
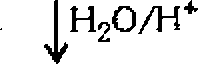 |
|
E |
CcHcNHCrO<>ei
(iv) chs(ch2)6ch2oh c\a2* F
{b) What is the pathway for the photolysis of acetone ?
Give various products formed. 4I
(c) Comment on the relative inertness of benzene
towards addition reactions. 2
6. (a) How are dyes classified on the basis of application ?
Give the method of preparation of an azo dye. 4!
(b) How is aspirin produced from phenol ? 2
(c) What are addition and condensation polymers ? Give
the preparation of one polymer of each kind. 4
(d) State Markownikoffs rule with suitable example. 2
CHE-6 4
|
Attachment: |
| Earning: Approval pending. |
