Visvesvaraya Technological University (VTU) 2007 B.E Chemical Engineering Chemical Engg Thermodynamics - Question Paper
Fourth sem B.E degree exam July 2007
Chemical Engg Thermodynamics
ISN
\W SCHEME
Fourth Semester fc E. Degree Examination, July 200
Ch .nical Engineering Chomlcal En( leering Thermodynamics
r>m:3hn.| {Max. Mark* IOO
Note ; Am*tr any h'll'fcfullquestiom.
I a. Lxptain
t) C'IomO ml open tyuem
ti> Adiabatic and itolaicd
tit) RcvcmNc md inevcrvhle process**
iv) tMthennal and aduhitic pnxcucf. (* wriM
b One mok of gti in a cIomxJ and wxvflow syitem un&rgocs a 4-ep ihernxxljtuaiK: cycle
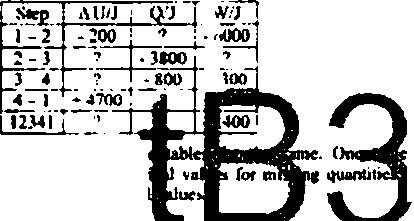
Cn*lnici two mi titilrBhmSnir Onelfle to *ho* e*juabom ued to calculate the mum iWt for tnrMnf ituontiticnnd the 2 table to write (He calculated nufnerkalflduefl m M
Show thei Energy baiafcetloMES!)' - ute JWroce for open tjncin is fives by.
AH* jA<r 0* Wj iliMfkw
A turbine operating under wcady Haw eooditioru receive* 5000 kf scam per hour The noun enter* at a vdociiy of 3000 m/mm, ai an elevation of 5 re and ipeciftc enthalpy 2777 kM* h learn the turbine at a \locit> of600 m'min and an elevation of 1 m and specific cnihal; y 2252 kJg Ileal toucs from turbine to urroundicig jnwunu io 16680 kJV$ D- rrruae HP output of the tu/binc. Now I HP 743.7 W
it) Marlul
Stale and prow nCmot Theorem' iijvmlm
Explain Lost Work" conccpi ( Mk*i
a. Sute anti prove CUutiut Inequality. Hi Mn*>
b. A heat pump i iaed to hctt a building dunng winter. The Knldtnf i W be maintained 21*C at all timev The btnkfang Use* heat at the rwe ofl J5000 IJ'h. when outtide temperature drop* to *5'C Determine minimum power required to
drive the beat pump (or lh <Kiti<ie temperature.
IM
IM
!*<
a. Explain the principle of entropy increatc h. Diicuts the 'Third Law of Therm odynamnV
t.HwJ 2
www.enggresources.com
I'age Au . 2 CH4J
k. a.*(i)r.(i)r.o
and
show that if du CvdT. Ii
(II MuiM
|
Derive Gtbbt-Duhem equation font funcUmc' 11k activity coefficient* data for a binary col arc cocretaied M. |
V (UMmIui at fixed ternperature and pressure |
|
2
Acclic acid i* e*tenfied tn the liquid phat according to CTIjCOOH (/) CjHjOll (ft CHjCOOC, The feed ccmi*U of 1.0 mo) of cach of acf fraction of elhyl acelale in the meting: mi*h> formation and standard free energy of formati* j CHiCOCHI il> C;H?OH <Q CT ' 2858101 237130'(
|
<UM<rlDi tnvnkii vith ethanol at 373 K and 1.0 bar i/>+!l|0<J> In- acid and ethanol Estimate the mote I equilibrium The Woodard heal of it 2?8 k are given below, fJOCiHj (/I 463250 318218 |
|
Assume that heal of reaction U iadcpcfuien ideal tollmen |
f temperature and Itqukl mixture is <l2V|k,| |
www.enggresources.com
174780
|
Attachment: |
| Earning: Approval pending. |


