Lovely Professional University 2010 B.Tech Electronics and Communications Engineering Chemistry Assignment - Question Paper
i m attaching paper
HOME WORK: 4
School: LSE Department: Chemistry
Name of the faculty member: Dr. Nisha Saxena Course No: CHE101 Course Title: Chemistry
Class: B.Tech. Term: I Section: B6004 & B6005 Batch: I
Max. Marks: 7 Date of Allotment: 9th Nov 2010 Date of Submission: 16th Nov 2010
PART A
Q.1 a) Discuss the acidic strength of following acids and arrange in decreasing order of their acidic strength.
i) FCH2COOH ii) ICH2COOH iii) BrCH2COOH iv) ClCH2COOH
b) CHCl3 is more acidic than CHF3 why?
Q.2 a) How many structural isomers are possible for C5H12? Draw structures and give their IUPAC name.
b) Give structures of α-chloropropanoic acid and β-chloropropanoic acid. Which one is more acidic and why?
Q.3 a) Give The IUPAC name for following compounds:
i)  ii)
ii)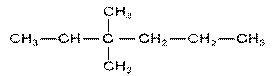
iii) ![]() iv)
iv) 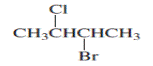
b) Draw the structure of following Compounds:
i) 2-propyl-4-methylhexane ii) 2-bromo-3-nitrobutane iii) 3,4,5-trimethyloctane
Part B
Q.4 a) Discuss the addition of HBr to unsymmetrical alkene (propene) in the presence of peroxide.
b) Arrange the free radicals (methyl radical, primary radical, secondary radical and tertiary radical) in increasing order of stability and discuss their stability on the basis of hyperconjugation.
Q.5 a) Give the mechanism of the following reaction:
(CH3)3C-Br + KOH (aq) → (CH3)3C-OH + KBr
b) How the nature of halogen atoms (leaving group) in alkyl halide play important role in SN2 reactions? Give the order of reactivity of alkyl halides having different halogen atom.
Q.6 a) Discuss the stereochemistry of polymers (isotactic, syndiotactic and atactic) by taking suitable example.
b) Identify the monomers in the following polymers and give their structure?
i) Polystyrene ii) Teflon iii) PVC iv) Nylon-66
| Earning: Approval pending. |
