Veer Narmad South Gujarat University 2010 B.E Engg.Chemistry - 2 - 2 ( Sem3 )( Chemical ) - Question Paper
RN-6123
B. E. - II (Sem. Ill) (Chemical) Examination May/June - 2010 Engg. Chemistry - II
Time : 3 Hours]
[Total Marks : 100
Instructions :
Seat No.:
M CnsLLnlcj.L{l [qoicu u? snqw <h>h41.
Fillup strictly the details of signs on your answer book.
Name of the Examination :
B. E. - 2 (Sem. 3) (Chemical)
Name of the Subject:
Engg. Chemistry - 2
|
-Subject Code No. |
|
-Section No. (1,2,.....) |
Student's Signature
1&2
(2) Question 1 and 4 are compulsory and carries 20 marks each.
(3) Question 2, 3 and 5, 6 carries 15 marks each.
(4) Answer to the two sections should be written in separate answer books with figures and mechanism wherever necessary.
(5) At wts. H=l, C=12, N=14, 0=16, S=32, Br=80, Cl=35.5, Na = 23, Ba = 137, Ag = 108.
(a)
10
(ii)
The boiling point of an organic liquids if available
in small quantity is usually determined by_
method.
In copper wire test the halogens presence in an
organic compound usually gives _ colour
flame.
Fill in the blanks (i)
(iii) The process of conversion of solid to gases directly on heating is called _.
(iv) A molecule is said to be chiral if it cannot be_
on it's mirror image.
(v) Diazotisation reaction can be effectively done with the help of_ and _ 0-5C.
RN-6123] 1 [Contd...
(vi) As per IUPAC name Acetamide is known as
(vii) Sulphanilamide is used as a sulpha drug due to its_ medicinal properties.
(viii) Criteria of purity of liquid is usually determined by its sharp _.
(ix) _isomeric aromatic hydrocarbon are possible
for C2H60.
(b) Answer the following : 10
(i) Explain in brief the combustion method for estimating carbon and hydrogen.
(ii) Explain Geometrical isomerism in detail.
2 Answer the following : (any three) 15
(i) Show the optical isomerism in lactic and tartaric acid.
(ii) Explain :
(a) Vacuum distillation
(b) Sublimation.
(iii) Write the preparation, properties and uses of :
(a) Acetaldehyde
(b) Phenol.
(iv) Write in detail about primary, secondary and tertiary alcohols.
3 Answer the followings : (any three) 15
(i) Write a note on :
(a) Steam distillation
(b) Fractional crystallisation.
(ii) Write the preparation, properties and uses of
(a) Citric acid
(b) a -naphthols.
(iii) Define isomerism. Write in detail about structural isomerism.
(iv) Explain the following qualitative test :
(a) Phthalic anhydride test for phenol
(b) Fehling test for aldehyde
(c) Mulikan barker test.
Fill in the blanks : 10
(i) Covalent bonds are formed by mutual_of
electrons.
(ii) Phenyl cyanide on acidic hydrolysis gives _.
(iii) Cummene process is used to prepare _.
(iv) Heterocyclic fission of an organic compound forms _ and _.
(v) As per IUPAC nomenclature the CH3COCl is name as _.
(vi) The full form of SN | reaction is _.
(vii) In reaction mechanism the attacking reagents are named as a _ and _.
(viii) Organic compounds consisting hetero atoms other than carbon are known as _ compounds.
Answer the following : 10
(b)
(i) Discuss the formation of Grignard Reagent and the precaution to be taken place within?
(ii) Explain electrophilic substitution reaction with mechanism.
5 Answer the following : (any five) 15
(i) Explain nitration with mechanism.
(ii) Write the penetration, properties and uses of salicylic acid.
(iii) Discuss what is peroxide effect and explain its importance in organic reactions.
(iv) Toluene uv light >A >B >c
Identify product A, B and C.
(v) Classify the following groups as O/P or m-directing in an aromatic nitration reaction :
(i) -CH3
(ii) -NH2
(iii) -Cl
(iv) -COOH
(vi) Write the preparation, properties and uses of resorcinol.
(vii) Write the qualitative tests to detect the presence of acid and alcohol.
6 Answer the following : (any three)
(i) Write the preparation (one), properties (two) and uses of (a) Cinnamaldehyde (b) Phloroglucinol.
(ii) Write a note on :
(a) Attacking reagent
(b) Mesomeric effect.
(iii) Write a note on different types of organic reactions.
(iv) Write the structural formula of the following :
(a) Citric acid
(b) Iodoform
(c) Acetophenone
(d) Thiophene
(e) Propyl methyl ether.
(v) Give the IUPAC nomenclature of the following structures :
(a) HOH2C - C = C - CH2OH
(b) H2C = C - CH = CH2
ch3
(c) H2C = CH - CO - CH3
(d)
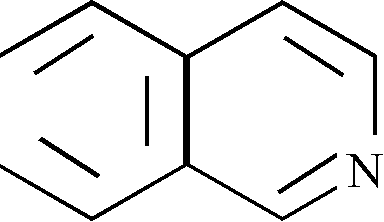
CH3
(e) CH. - CH, - C - COOH
CH,
RN-6123] 4 [ 200 ]
|
Attachment: |
| Earning: Approval pending. |
