Indian Institute of Technology Roorkee (IIT-R) 2009-2nd Year B.Tech Biotechnology Principles of bioreaction engineering, - Question Paper
Department of biotechnology
Indian Institute of Technology Roorkee
BT 215 Principles of bioreaction engineering
End term exam Autumn semester 2009-2010
Summary:
This ques. paper covers elementary topics on reaction engineering like mass balance, energy balance , enzyme kinetics and reactor designs.
: of Biotetfmoiocy tasfcar* tnstibitt of Tecfrmotogy Roorkee
ST 21S P. kcjyries oi &'orection Engineering '
EC Tenx Ennwaton, Autumn Semester 2009*2010
MMkwwilteteiae____Tlmr. 3 houri
-VTS*er A,!.' Qjesttxs
1. How nvinji pctircs cf jcduni suitpihte v? tiyjfft!) to remove the oxygen from 8330000 lb of *cer contamrg 10 pcrrr of or*e*i aid * the same tme maintain a 35% excess of sodium sufcht*' 7
|
2. For ptot ptiffc cuc'inea i&ire I tte to/hr of the jecyde stream R? All are m __e *7 0 |
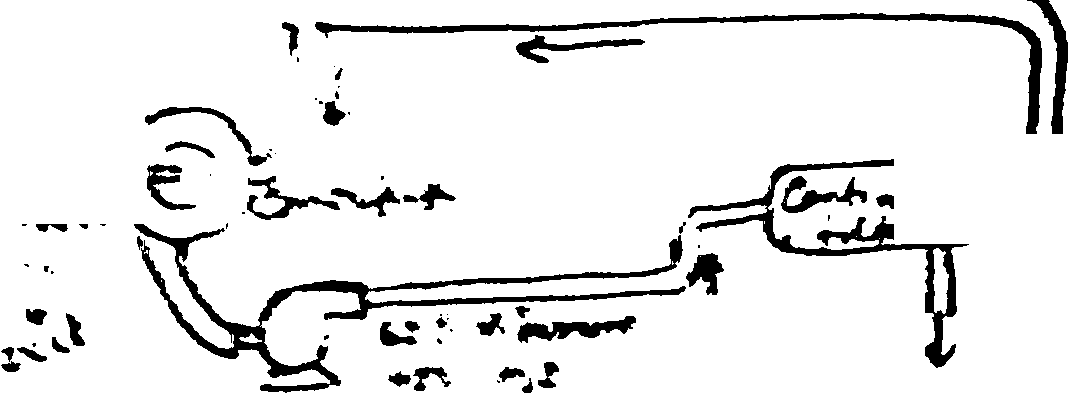 |
} fmt*.*- itoLaor"
, - irf? *'**5-*
VVvA -
i. Air Nie cc<Ttcressn5 *ctt IX ca.. 2X cf*rc%aipf 429 kJ/kgj to 1000 kPa and 278 K (enthalpy 509 H Vg.'. the npvjc m*s 2 i t* **vc s r*e et velocity of the air from compressor if the
baa q IX $ h of 7
4. Heat cacaolv iica v jr-TP-crna 2s s g nw wr F* an equation of the form Cp * a * bT + cT2
| |||||||||||||||||||||||||
|
13 |
S. Ho* mans \andtoe-s irxst ae spectiec *or tSe fjmace shown below (figure 2)t0 absorb all
degrees of fr*e&x~i:
10
I 1 n'f
<*v
Co
w
|
Tu/i 7*V |
fuu. | |
|
rivijvu 4 - |
1 UtwF |
I &0r*)
*1,
a i rTR* ire available, with a volume ot 8 and 2
6. For the production of B from A two ideal cs concentration of 8 kmol/m A.
respectively. The feed is a stream (4 mW) of Aw.a concen # #
The reaction proceeds according to A . vai,ie is expected for the reaction order,
small ideal CS TR at the same temperature. A fractional value is expected
|
The following observations were made: | |||||||||||||||||||||||||||
|
What is the maximum possible yield of B in the practical situation?
. 7. Reactant A in a liquid either isomerizes or dimerizes as follows A Rdcts/ed rR R|Ca
A A 1*S 2
a) Write expression for fractional yield ,vj/ (r/r+s>
b) With a feed stream concentration of Cao find CRmaX which can be formed in a plug flow reacior arid in a mixed flow reactor.
8. Show optimum temperature progression profiles for adiabatic operation of reversible endothermic and exothermic reactions in a plug flow reactor and method to obtain the volume of reactor for these t>pe reactions in a plug flow.
9. A new reactor has been installed for the production of B by the first order irreversible reaction 2 A ->B. Because it is presumed that the reactor is not ideal, the non-ideal behavior is investigated using a pulse marker. At the exit of the reactor the following marker concentrations are measured, as response to a pulse at t = 0 at the inlet:
Time [mm] 10 20 30 40 50 60 70 SO C [mol liter] Q 3 5 5 4 2 10
Plot the age distribution curve, E curve for this reactor. Based on these measurements, which conversion can be expected in the new reactor, if it is known that in an ideal CSTR at the space time chosen for the nc reactor, a conversion of 80% is achieved?
(8+7)
|
Attachment: |
| Earning: Approval pending. |
